Solving a medical mystery and changing CDC screenings for COVID-19
What we learned from the first known community-acquired coronavirus case in the U.S.
When UC Davis announced the first case of community transmission of COVID-19 in the U.S. on Feb. 26, it solved a medical mystery at the hospital and led to important changes to the U.S. Centers for Disease Control and Prevention (CDC) guidelines for novel coronavirus testing.
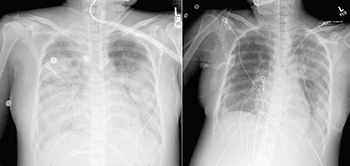
In a paper published today in Clinical Infectious Diseases, UC Davis Health physicians and medical staff who treated the severely ill patient provide a detailed case study of her condition and the medical steps and challenges they experienced before arriving at a diagnosis and treatment. The case study also reveals how her symptoms matched—and sometimes varied from—published studies of COVID-19 infection at the time.
SARS-CoV-2, the novel coronavirus that causes COVID-19, was first identified in a small number of cases in Wuhan, China in December 2019. As of March 30, the virus has infected over 752,830 people around the globe and caused more than 36,230 deaths. In the U.S., the CDC reports 140,904 have tested positive with 2,405 deaths. In California, the California Department of Public Health reported 4,643 positive cases and 101 deaths.
No known risks for novel coronavirus
An otherwise healthy woman in her 40s, the patient was admitted to UC Davis with a respiratory infection. Her chest imaging suggested community acquired pneumonia. The patient was immediately placed on droplet and contact precautions to prevent infection transmission.
Within 24 hours of admission, her respiratory status deteriorated. She was intubated and given antibiotics including linezolid, piperacillin-tazobactam and azithromycin. Testing over several days—viral panel, respiratory culture, blood cultures, bronchoscopy cultures—failed to indicate a clear infectious source.
The UC Davis team suspected a potential COVID-19 infection. However, because the patient had not traveled to high-risk countries (at the time China) and had no contact with an individual with high-risk travel, she did not meet the CDC criteria. As a result, public health officials did not pursue testing.
The patient developed acute respiratory distress syndrome, a condition in which fluid builds up in the lungs and limits the oxygen that can reach the bloodstream. She developed a septic shock, a potentially fatal sharp drop in blood pressure in reaction to severe infection.
Because of the severity of her respiratory condition, the patient was reviewed again for possible coronavirus infection. This time, the CDC recommended COVID-19 testing. The patient was put on airborne precautions and strict contact precautions. Two days later, the results came back positive.
No approved therapies for the novel coronavirus
Currently, there are no approved antiviral therapies for COVID-19 in humans, but clinical trials are underway at several academic medical centers, including UC Davis. Because of the severity of the patient’s illness, the team received approval from the Food and Drug Administration (FDA) to treat her with an investigational drug called remdesivir (brand name Veklury). The broad-spectrum antiviral developed by Gilead Sciences Inc., has been tested in humans with Ebola and has shown promise against coronaviruses in animal models.
After receiving remdesivir infusions, the patient improved, needing significantly less ventilator support and having better blood oxygen levels and chest X-ray results. Fourteen days after first arriving at UC Davis, she was removed from mechanical ventilation. She has since been discharged and is recovering at home.
The team emphasizes that whether remdesivir is effective against human COVID-19 is not yet known. Clinical trials funded by the National Institutes of Health (NIH) and the pharmaceutical industry will be key to analyzing the drug’s efficacy against SARS-CoV-2.
“Given the urgent need to find an effective treatment for COVID-19, clinical trials are essential for determining, from a scientific standpoint, if remdesivir is safe and effective,” said Allison Brashear, dean of the UC Davis School of Medicine. “With this new study funded by the NIH, UC Davis will be an important contributor to these critical efforts.”
Testing is key to tackling pandemic
The case highlights significant knowledge gaps in the diagnosis and management of COVID-19. Without clear risk factors, the patient’s infection first masqueraded as a community acquired pneumonia. She also was relatively young and without other health conditions that would identify her as at-risk for severe disease.
“Our case has influenced national health policies for revising screening criteria,” said Angela Haczku, associate dean for translational research at the UC Davis School of Medicine and senior author on the study.
Because of this specific case, and other similar cases of community-acquired COVID-19, the CDC updated its guidelines so that any hospitalized patient with severe symptoms, such as acute respiratory distress syndrome or pneumonia without an explanatory diagnosis, can now be tested for COVID-19 even if no clear source of exposure is identified.
“There are individuals in the community who are not manifesting severe enough symptoms to check with their health care providers,” said Michael Schivo, co-director of UC Davis Comprehensive COPD Clinic and senior author on the study. “We expect community spread to occur more frequently, challenging the ability of health care systems to adequately contain the spread of COVID-19.”
As the virus continues to spread, and more data about it becomes known, the authors expect guidelines to change yet again. But they are adamant that to tackle the pandemic, there needs to be significantly faster, less expensive and more widespread testing of all patients who potentially have COVID-19.
Other UC Davis Health researchers on the study “A community transmitted case of acute respiratory distress syndrome due to SARS-coV2 in the United States” include Stuart Cohen, Bradley Sanville, Rebecca Corbett, Wesley Pidcock, Kaitlyn Hardin, Christian Sebat, George Thompson III and Minh-Vu Nguyen. The clinical trial is being funded by the National Institute of Allergy and Infectious Diseases (NCT04280705).



