Jiejun Wu, MD/PhD; Samer Albahra, MD; Nam K Tran, PhD
Introduction: History and clarification of some nomenclatures
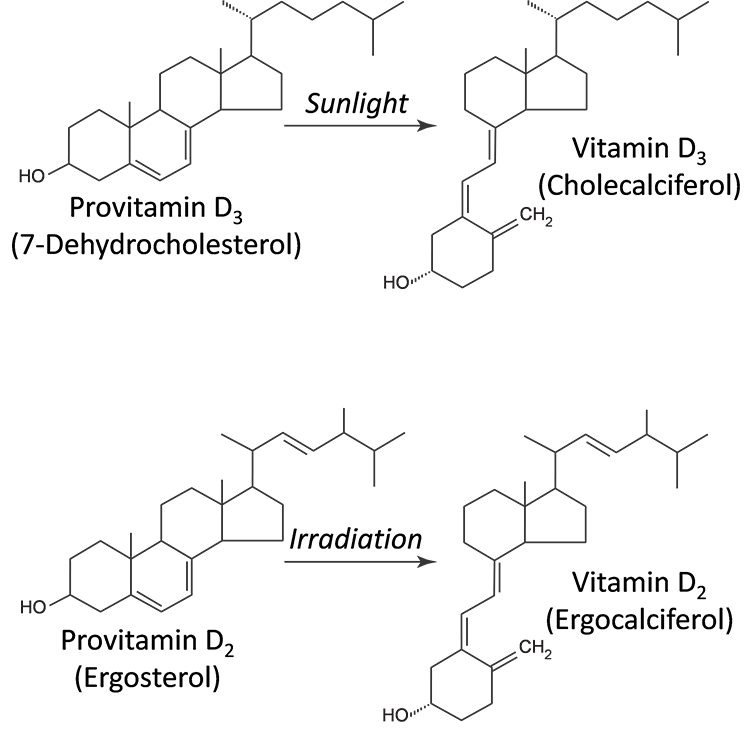
The discovery and importance of vitamin D was first revealed in 1919 when Sir Edward Mellanby successfully reproduced a nutritional disease known as rickets by feeding dogs oatmeal and keeping these dogs from exposure of ultraviolet light. This mystery vitamin was then termed as “vitamin D”. 1 This term, however, does not differentiate between vitamin D3 (cholecalciferol) of human or animal and vitamin D2 (ergocalciferol) of plant origin (Figure 1). 7-dehydrocholesterol also referred to as pro-vitamin D3, is naturally occurring and abundantly available in the skin epidermis, probably as a side product from cholesterol synthesis. Under sunlight (solar ultraviolet radiation at 290-315 nm), D3 is synthesized from precursor 7-dehydrocholesterol. For human beings, another source of D3 is animal foods. Dietary D2 may be obtained from plant source and often found in commercially manufactured nutrition additive by irradiating provitamin D2, ergosterol. Both D3 and D2 are equally potent and biologically equivalent but D3 is the dominate form in humans.2-4
Structure and Functions of Vitamin D
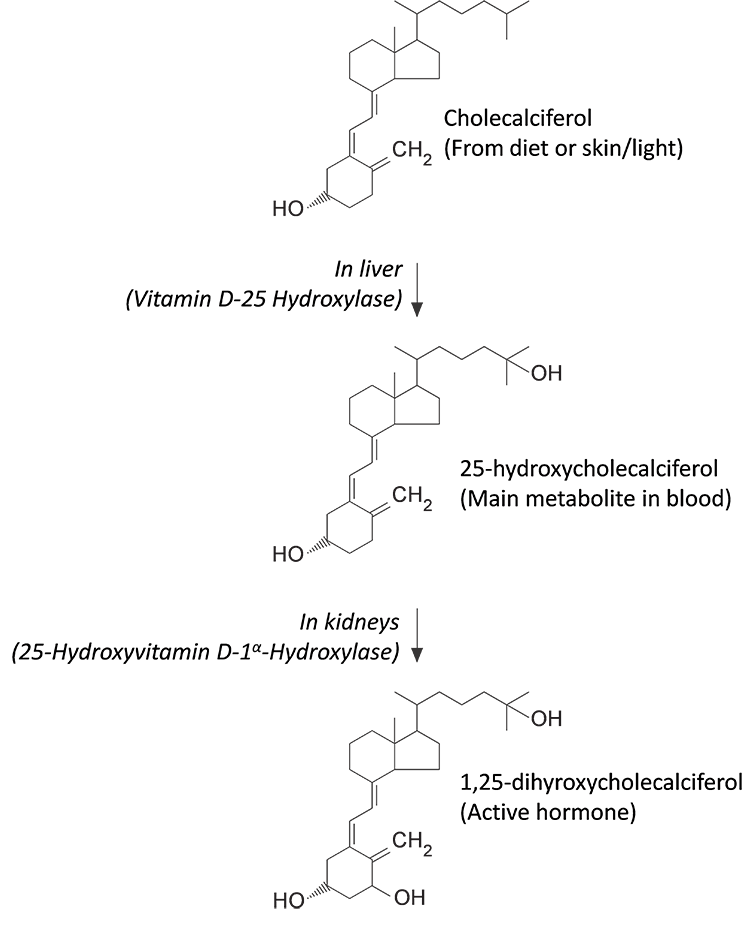
Vitamin D obtained either from skin exposure to sun slight or from food is inactive. It is either stored in adipose tissue or further converted to 25-hydroxyvitamin D (25-hydroxycholecalciferol) in the liver by the action of an enzyme called vitamin D-25 hydroxylase. 25-hydroxyvitamin D is not the biologically active form and requires another hydroxylation step to increase its activity (1000x increase in activity) forming 1,25-dihydroxyvitamin D (1,25-dihydroxycholecalciferol). This hydroxylation step is completed in the kidney (proximal tubular epithelial cells) with the help of 25-hydroxyvitamin D-1α-Hydroxylase (Figure 2).2-5
In bone metabolism, the plasma concentrations of calcium and phosphate are regulated by three hormones, primarily parathyroid hormone (PTH) and Vitamin D, and to a lesser extent by calcitonin. 1,25-dihydroxyvitamin D plays a major role in maintaining calcium and phosphate concentrations by its actions on intestine, bone, kidney and the parathyroid glands. It increases the absorption of calcium and phosphate in the small intestines, promotes the maturation of osteoclasts and bone resorption, negatively regulate its synthesis in the kidney and inhibit the production of parathyroid hormone in the parathyroid. As a steroid hormone, 1,25-dihydroxyvitamin D performs its biological activity by binding its nuclear acceptor to regulate target genes’ expression.2
Beyond its classic function on bone metabolism, more and more evidence suggest that vitamin D could be an important regulator in immune response and epithelial differentiation. A low level of vitamin D has been associated with a range of malignancies, autoimmune diseases, cardiovascular and infectious diseases, and recently, COVID-19 which will discussed in more details below.6
Laboratory Best Practice of Vitamin D status
There is an increasing demand on assessment of vitamin D status however, the definition of vitamin D sufficiency has been controversial. This is mainly caused by the poor correlation between blood concentration of the vitamin and its associated clinical sequela which is disturbed by parathyroid hormone and another bone-derived hormone fibroblast growth factor-23, and likewise population differences also exist. These confounding factors ultimately create a discordance between reference intervals and testing cut-offs cited in literature. In brief, modern vitamin D assays are now being standardized under the Vitamin D Standardization Program (VDSP) and certified by the Centers for Disease Control and Prevention (CDC). The difference of reference intervals and cut-offs should be discussed since it is often confused by healthcare professionals. All tests, not just vitamin D, define reference intervals based on a “reference population” of apparently normal individuals. Often, these reference populations are derived from diverse cohorts of a few hundred individuals and can differ between one manufacturer to the next. As such, some studies have suggested population specific vitamin D cut-offs may be more appropriate.9 In contrast, cut-offs are defined by scientific/clinical data or through consensus. The current recommended 25-hydroxyvitamin D cut-off for sufficiency ranges 20-30 ng/mL (50-75 nmol/L) depending on sources. Deficiency is defined as 25-hydroxyvitamin D levels of less than 10 ng/mL (25 nmol/L). Levels between 10-20 ng/mL (25-50 nmol/L) are considered suboptimal for bone health and termed as insufficient. Vitamin D intoxication is rare, with 25-hydroxyvitamin D concentration higher than 150 ng/mL (375 nmol/L) as more likely to cause toxicity.7 Interestingly, studies evaluating vitamin levels in “apparently healthy” populations with adequate sun exposure have shown vitamin D levels can be as low as 15 ng/mL which ultimately can influence reference intervals for any assay.8
The half-life of active vitamin D, 1,25-dihydroxyvitamin D is only four hours (Table 1) while 25-hydroxyvitamin D is more stable reflecting blood vitamin D levels and selected for testing.2 However, this may cause issues in patient with renal disease when 25-hydroxyvitamind D does not represent the active metabolite of vitamin D.
Table 1 The half-lives of vitamin D metabolites in plasma
| Compound | Concentration in Plasma | Half-Lef |
| Vitamin D | <0.2-20 ng//mL <0.5-52 nmol/L |
1-2 days |
| 25-Hydroxyvitamin D | 10-65 ng//mL 25-162 nmol/L |
2-3 weeks |
| 1,25-Dihhydroxyvitamin D | 15-60 pg//mL 36-144 pmol/L |
4-6 hours |
Immunoassay or mass spectrometric methods are the methods used in vitamin D level testing. Liquid chromatography with tandem mass spectrometry (LC-MS/MS) serves as the reference method but requires expensive instruments and can be time consuming, therefore, not routinely used in most facilities. Immunoassay is a popular alternative technique which are standardized to LC-MS/MS methods. Recently, an automated sandwich-type immunoassay was developed commercially for 25-hydroxyvitamin D with good sensitivity and specificity and high correlation with spectrometric method.4 At UC Davis Health, vitamin D testing is performed using an VDSP/CDC certified immunoassay method, as such vitamin D cut offs can remain the same. Performance comparison of the old versus new vitamin D assay is summarized in Table 2.
Table 2. Comparison of old (Beckman) versus new (Roche) vitamin D assays.
| Beckman Vitamin D | Roche Elecsys Vitamin D Total II | |
| Method | CLIA | ECLIA |
| Accuracy | Both methods have been standardized by the manufacturer against LC-MS/MS 25-hydroxy vitamin D reference method procedure using donor patient sample from the Vitamin D Standardization Certificate Program. Comparisons showed no statistically significant difference between the two methods at UC Davis Health. | |
| Precision (CV) | ||
| 15 ng/mL | 4.6% | 4.8% |
| 25 ng/mL | 4.7% | 3.0% |
| 50 ng/mL | 3.0% | 1.6% |
| 100 ng/mL | 3.0% | 1.9% |
Abbreviations: CLIA, chemiluminescent immunoassay; CV, coefficient of variation; ECLIA, electrochemiluminescent immunoassay.
Vitamin D and COVID-19
The depletion of lymphocytes and the increase of proinflammatory cytokines such as IL-6, is believed to be the central pathophysiological process causing multisystemic damage in COVID-19 patients, and this immune dysfunction is associated with the increased severity and mortality of this disease.10-11 In adults, this acute systemic inflammatory syndrome is called cytokine release syndrome12; In children, this Kawasaki-like hyper-inflammation is termed multisystem inflammatory syndrome in children.13
There are more and more publications showing non-skeletal functions of vitamin D, especially its inhibitory effects on the production of pro-inflammatory cytokines including IL-6, and the association of its deficiency with many immune-related diseases.14 One of such disease is COVID-19.7,15-16 A recent work investigated pre-infection 25-hydroxyvitamin D3 levels and its association with severity of COVID-19 illness and showed that vitamin D deficient patients has statistically significant higher mortality (25.6%) than vitamin D sufficient group (25.6% vs 2.3%, p = 0.001).17 Therefore, this is a relevant topic that deserves more attention and further investigation. However, the current data, mostly from retrospective studies, have not established a definite cause-effect relationship between vitamin D levels and its suggested anti-COVID-19 efficacy; further investigation, such as well-controlled clinical trials, may be needed before vitamin D could be considered in prevention and clinical management of COVID-19 cases. One thing needs to be mentioned is that, before more evidence is available, routine screening of vitamin D is currently not recommended since, as mentioned earlier, clinical interpretation of this testing result is often complicated and does not change clinical management.7,18
References
- Deluca HF. The Genetics and Biology of Vitamin D. in Principles of Medical Biology, 1997
- Fraser WD. Fraser. Bone and Mineral Metabolism. in Tietz Textbook of Clinical Chemistry and Molecular Diagnostics, 2018
- Dasgupta A, Wahed A. Clinical Chemistry, Immunology and Laboratory Quality Control A Comprehensive Review for Board Preparation, Certification and Clinical Practice 2014.
- McPherson RA, Pincus MR. Henry's Clinical Diagnosis and Management by Laboratory Methods. 2021.
- Harvey R, Ferrier D. Lippincott Illustrated Reviews: Biochemistry. 2011
- Prietl B, et al. Vitamin D and immune function. 2013 Jul 5;5(7):2502-21
- US Preventive Services Task Force, et al. Screening for Vitamin D Deficiency in Adults: US Preventive Services Task Force Recommendation Statement. JAMA. 2021 Apr 13;325(14):1436-1442.
- García-Dorta A, et al. Association of Gender, Diagnosis, and Obesity with Retention Rate of Secukinumab in Spondyloarthropathies: Results Form a Multicenter Real-World Study. Front Med (Lausanne). 2022 Jan 13;8:815881
- Hilger J, et al. A systematic review of vitamin D status in populations worldwide. Br J Nutr. 2014 Jan 14;111(1):23-45.
- Liu J, et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. 2020 May;55:102763.
- Liu F, et al. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. 2020 Jun;127:104370.
- Gubernatorova EO, et al. IL-6: Relevance for immunopathology of SARS-CoV-2. Cytokine Growth Factor Rev. 2020 Jun;53:13-24
- Feldstein LR, et al. Multisystem Inflammatory Syndrome in U.S. Children and Adolescents. N Engl J Med. 2020 Jul 23;383(4):334-346
- Charoenngam N, Holick MF. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients. 2020 Jul 15;12(7):2097
- Jain A, et al. Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers. Sci Rep. 2020 Nov 19;10(1):20191.
- Meltzer DO, et al. Association of Vitamin D Status and Other Clinical Characteristics With COVID-19 Test Results. JAMA Netw Open. 2020 Sep 1;3(9):e2019722.
- Dror AA, et al. Pre-infection 25-hydroxyvitamin D3 levels and association with severity of COVID-19 illness. PLoS One. 2022 Feb 3;17(2):e0263069.
- Levy E, Delvin E, Vitamins: functions and assessment of status through laboratory testing. In Contemporary Practice in Clinical Chemistry. 2020.



