Resident Program - Case of the Month
March 2020 - Presented by Dr. Anupam Mitra (Mentored by Dr. John Paul Graff)
Clinicopathologic Features of Burkitt Lymphoma and Differential Diagnosis
Background
Burkitt lymphoma is a highly aggressive but curable lymphoma originally identified and described by Dr. Denis Burkitt in Africa while working as a surgeon there during the mid-nineteen hundred. He observed that large tumor masses were distorting young children’s faces, specifically the jaw and mandibular regions. Some of these patients even had abdominal masses but without a discernable lymphadenopathy. Previously misinterpreted as a sarcoma, [1, 2] these entities were later established to be a lymphoma and given the name Burkitt lymphoma after Dr. Burkitt. This lymphoma was found to be the most common tumor in tropical Africa with an exception being Africans who lived at higher altitudes or in cooler climates. [2] These findings were puzzling for Dr. Burkitt. As a result, he collaborated with Dr. Epstein and his colleagues to find if any association existed between this disease and a virus known as Epstein-Barr virus (EBV). They were successful in identifying the association of EBV virus in the pathogenesis of Burkitt lymphoma. [2] Although very limited information was available at that time, yet these patients did exceedingly well with surgery or chemotherapy. [2]
Pathologic Features
In the current World Health Organization (WHO) Classification (2016), [3] three epidemiological variants of Burkitt lymphoma are described: endemic, sporadic, and immunodeficiency-associated types. These clinical variants differ not only in their geographical distribution, but also in their clinical presentation, molecular genetics and biological features.
Endemic Burkitt lymphoma occurs in malaria prone areas of equatorial Africa and Papua New Guinea. It involves children, usually 4–7 years old, with a male to female ratio of ~2:1. The facial bones, as well as kidneys, gastrointestinal tract, ovaries, breast, and other extra nodal sites (thyroid, salivary glands) are the most common sites of involvement. [4] The frequency of Burkitt lymphoma in these areas is estimated to be 50 times higher than in the U.S. [5]. EBV is found in nearly all cases.
Sporadic Burkitt lymphoma occurs worldwide and has no specific geographic or environmental association. It accounts for 1–2% of lymphomas in adults and up to 40% of lymphoma in children in the U.S. and western Europe. [5] The median age is 30 years, with a male to female ratio of ~2-3:1 and has an increased incidence in the elderly population. Unlike endemic Burkitt, sporadic Burkitt rarely involves the face. The abdomen, especially the ileocecal area is the most common site of involvement; the ovaries, kidneys, omentum, and other sites may also be involved. Bilateral massive involvement of the breasts may occur in association with the onset of puberty or with lactation. [5] Lymph node involvement is rare but more commonly seen in adults than in children. [6]
Immunodeficiency-associated Burkitt lymphoma occurs primarily in patients infected with HIV but can also be seen in allograft recipients [7, 8] and individuals with congenital immunodeficiency. Compared with HIV positive patients with diffuse large B-cell lymphoma, those with Burkitt lymphoma are younger, less often carry a prior diagnosis of AIDS, and have higher mean CD4 counts (usually >200 cells/μl). The diagnosis of Burkitt lymphoma in an HIV positive individual often represents an AIDS-defining criterion. [9, 10]. HIV-associated Burkitt lymphoma presents with nodal disease and bone marrow involvement. Burkitt lymphoma occurring in transplant recipients tends to occur after a relatively long post-transplant interval (~4.5 years in one series). [7] Most patients are solid organ recipients, however recipients of stem cells transplant may be affected as well, though at a much lower incidence. Like endemic Burkitt, EBV is commonly seen but not uniformly present. [7, 8]
Histology and immunophenotype
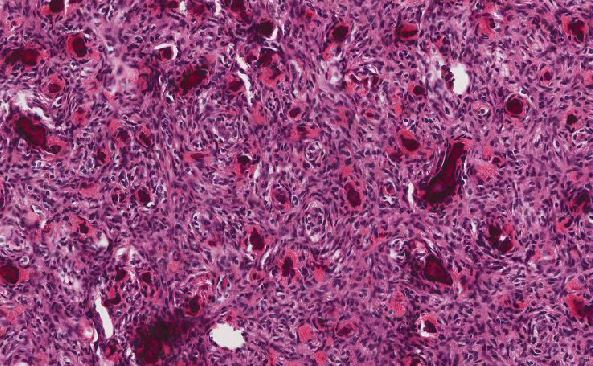
The tumor cells are intermediate in size with diffuse monomorphic growth. The nuclei are round with finely clumped chromatin and contain multiple nucleoli. The cytoplasm is deeply basophilic and usually contain lipid vacuoles (as seen in our case, Figure 4). The tumor has a very high proliferation index >95% by Ki-67 (as seen in our case, Figure 3) or mitotic count. It also has a high cell turnover with an elevated apoptotic rate, which creates the “starry-sky” appearance through the presence of multiple tingible body macrophages. The tumor cells generally express strong surface immunoglobulin (i.e. kappa or lambda), not noted in our case (flow cytometry was likely negative due to marrow heterogeneity and sampling). The cells are also positive for B cell antigens like CD20, CD22, CD79a, PAX-5 and germinal center markers like CD10 and BCL-6. Almost all cases of Burkitt lymphoma have strong expression of CMYC (as seen in our case, Figure 6). The characteristic lipid vacuoles in the cytoplasm can be highlighted using a lipid immunostain like adipophilin or oil red O. Due to its high apoptotic rate, this tumor is typically negative for BCL-2, which is an anti-apoptotic marker.
Genetic Profile
The characteristic molecular finding of Burkitt lymphoma is the translocation of myc gene (at chromosome 8) to the IgH (immunoglobulin heavy chain) gene (at chromosome 14) or less commonly to the IgK (immunoglobulin kappa) gene (at chromosome 2) or the IgL (immunoglobulin lambda) gene (at chromosome 22). [3] Approximately 10% of the Burkitt cases can lack myc gene rearrangement. Additional chromosomal abnormalities occurring in Burkitt include gains of 1q, 7 and 12 and loss of 6q, 13q32-34 and 17p. The WHO has also proposed that Burkitt with 11q alteration be classified as a sperate but related entity.
Differential diagnosis
The main differential diagnosis of Burkitt lymphoma are other types of high-grade B-cell lymphoma, including diffuse large B-cell lymphoma (DLBCL). [11] This problem is most relevant for adults rather than children as a much higher proportion of non-Hodgkin’s lymphoma in adults are DLBCL. The usual immunophenotypic pattern of Burkitt (as stated above) with its characteristic translocations for MYC can help in differentiating it from DLBCL, which are generally CD20+, CD10−/+, monotypic sIg+/− and have more commonly BCL-2 and BCL-6 abnormalities with or without MYC abnormalities. Furthermore, DLBCL’s neoplastic lymphocytes are medium to large as compared to Burkitt with small to intermediate cell size.
Similarly, in children, precursor B-lymphoblastic lymphomas (B-ALL) should be considered in the differential. [12] These patients generally present more commonly with circulating lymphoblasts (leukemia) than with tissue involvement. Moreover, these cells are small to medium in size and are polymorphic. Generally, these precursor B-ALLs don’t have a MYC rearrangement and the cells are typically CD19, CD10 and TDT positive without surface immunoglobulin expression and have diminished or no expression of mature B cell marker like CD20, kappa or lambda.
Another entity causing a diagnostic dilemma is blastoid variant of mantle cell lymphoma. These are seen mostly in middle-aged and older adults, male > female; and usually presenting as widespread disease in nodes and extra nodal sites. The cells are medium-to-large sized lymphoblast-like, pleomorphic cells with scant cytoplasm. There are typically no cytoplasmic vacuoles. These cells generally express CD20+, CD5+, CD10−, BCL-6−, BCL-2+, SOX-11+, cyclin D1+(BCL-1), with surface immunoglobulin and have a characteristic translocation, t(11;14) involving the bcl-1 (cyclin D1) and IgH (immunoglobulin heavy chain) genes.
Prognosis and predictive factors
Burkitt lymphoma is highly aggressive but potentially curable tumor. Overall survival is up to 90% with modern chemotherapeutic regimens. Adverse prognostic factors include advanced stage, bone marrow or central nervous system involvement, tumor size exceeding 10 cm, and high serum lactate dehydrogenase levels. [3] Relapse can occur rarely within the first year of diagnosis.
References
- Burkitt D. A sarcoma involving the jaws in African children. Br J Surg. 1958;46(197):218-23.
- Burkitt DP. The discovery of Burkitt's lymphoma. Cancer. 1983;51(10):1777-86.
- Swerdlow S CE, Harris N, Jaffe E, Pileri S, Stein H et al. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Revised 4th ed. ed: Lyon: International Agency for Research and Cancer (IARC; 2017.
- J. D. Burkitt lymphoma. Jaffe E HN, Stein H et al., eds., editor. Washington, DC: IARC Press; 2001.
- Blum KA, Lozanski G, Byrd JC. Adult Burkitt leukemia and lymphoma. Blood. 2004;104(10):3009-20.
- Boerma EG, van Imhoff GW, Appel IM, Veeger NJ, Kluin PM, Kluin-Nelemans JC. Gender and age-related differences in Burkitt lymphoma--epidemiological and clinical data from The Netherlands. Eur J Cancer. 2004;40(18):2781-7.
- Gong JZ, Stenzel TT, Bennett ER, Lagoo AS, Dunphy CH, Moore JO, et al. Burkitt lymphoma arising in organ transplant recipients: a clinicopathologic study of five cases. Am J Surg Pathol. 2003;27(6):818-27.
- Xicoy B, Ribera JM, Esteve J, Brunet S, Sanz MA, Fernandez-Abellan P, et al. Post-transplant Burkitt's leukemia or lymphoma. Study of five cases treated with specific intensive therapy (PETHEMA ALL-3/97 trial). Leuk Lymphoma. 2003;44(9):1541-3.
- Knowles DM. Etiology and pathogenesis of AIDS-related non-Hodgkin's lymphoma. Hematol Oncol Clin North Am. 2003;17(3):785-820.
- Martinez-Maza O, Breen EC. B-cell activation and lymphoma in patients with HIV. Curr Opin Oncol. 2002;14(5):528-32.
- Frost M, Newell J, Lones MA, Tripp SR, Cairo MS, Perkins SL. Comparative immunohistochemical analysis of pediatric Burkitt lymphoma and diffuse large B-cell lymphoma. Am J Clin Pathol. 2004;121(3):384-92.
- Bowman WP, Shuster JJ, Cook B, Griffin T, Behm F, Pullen J, et al. Improved survival for children with B-cell acute lymphoblastic leukemia and stage IV small noncleaved-cell lymphoma: a pediatric oncology group study. J Clin Oncol. 1996;14(4):1252-61.

 Meet our Residency Program Director
Meet our Residency Program Director