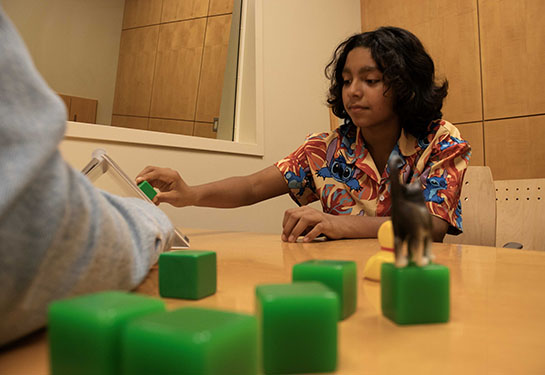First-ever in-utero stem cell therapy for fetal spina bifida repair is safe, study finds
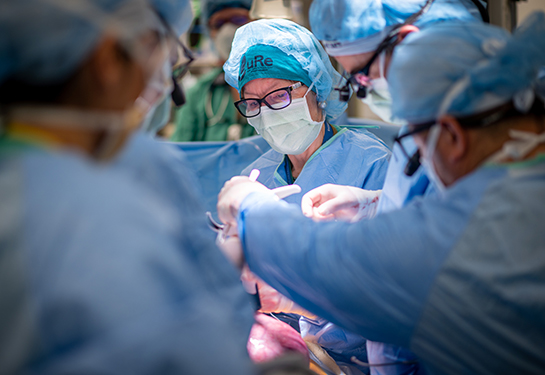
A UC Davis Health research team has safely performed the world’s first spina bifida treatment combining fetal surgery with stem cells, according to results from Phase 1 of an ongoing clinical trial. These results have been published today in The Lancet.
The study, called “Feasibility and Safety of Cellular Therapy for In-Utero Repair of Myelomeningocele (CuRe Trial): a First-In-Human, Phase 1, Single-Arm Study," tested whether adding a layer of human placenta-derived stem cells to standard fetal surgery could be done safely. Spina bifida, also known as myelomeningocele, occurs when spinal tissue fails to fuse properly during the early stages of pregnancy. The birth defect can lead to a range of lifelong cognitive, mobility, urinary and bowel disabilities.
“Putting stem cells into a growing fetus was a total unknown. We are excited to report great safety,” said Diana Farmer, the CuRe Trial’s principal investigator and chair of the UC Davis Department of Surgery. “It paves the way for new treatment options for children with birth defects. The future is exciting for cell and gene therapy before birth.”
The first phase of the trial was funded by a $9 million state grant from the state’s stem cell agency, the California Institute for Regenerative Medicine (CIRM).
About the treatment
During fetal surgery, a small opening is made in the uterus. The surgeons then float the fetus up to that incision point so they can expose the back of the fetus and the spina bifida defect. The care team places a small patch containing living stem cells directly over the fetus’s exposed spinal cord, before closing the layers of the back to allow the tissue to regenerate. The stem cells, taken from donated placentas, are designed to protect the developing spinal cord from further damage before birth.
It is the world’s first in-utero stem cell therapy for spina bifida, and the only trial aimed at improving the outcomes for the children over fetal surgery alone.
Key Phase 1 findings
The first six babies in the trial were monitored closely from surgery through birth. Researchers reported:
- No safety concerns related to the stem cells.
- There were no infections, spinal fluid leaks or abnormal tissue growth or tumors formed at the repair site.
- All surgeries were successful, and the stem cell patch was placed as planned for every patient, and all the wounds were completely healed.
- MRI scans showed reversal of hindbrain herniation in all infants, which is an indicator of surgical success.
- No babies required a shunt for hydrocephalus before hospital discharge.
Because the early safety results were strong, the Food and Drug Administration and an independent monitoring board approved moving forward with the next phase of the study.

Why this matters
Spina bifida affects 1,500 to 2,000 children in the United States every year. While fetal surgery has greatly improved outcomes since it was introduced more than a decade ago, many children still struggle with mobility and have other long-term complications. The CuRe Trial is exploring whether stem cells can add regenerative power to surgery, potentially improving mobility and quality of life.
“This is a major step toward a new kind of fetal therapy, one that doesn’t just repair but potentially helps heal and protect the developing spinal cord,” said Aijun Wang, co-inventor of the placental-derived stem cell treatment technology and the study’s co-principal investigator with Farmer. He is co-director of the UC Davis Center for Surgical Bioengineering.
For CuRe Trial participant Michelle Johnson, the decision to participate in the trial was life changing.
"Our family couldn't feel more blessed and fortunate to participate in the CuRe Trial,” said Johnson, whose son Tobi was born in 2022. “Tobi's physical and mental abilities are nothing short of a miracle. We are forever grateful for the many health professionals who supported Tobi's journey and continue to watch him conquer the world.”
Next steps
The CuRe trial is now enrolling up to 35 patients in its Phase 1/2a study. Children will be followed through age 6 to evaluate long-term safety and early signs of improved movement, bladder and bowel function.
The Phase 1/2a study is funded by CIRM and Shriners Children’s.