Ryan Thomas, MD; Samer Albahra, MD; Scott Bainbridge, CLS; Nam K Tran, PhD
Introduction
The current pandemic has led to considerable interest in COVID-19 related laboratory testing. The widespread distribution of the various vaccines, the emerging genetic variants of SARS-CoV-2 circulating around the world, and their impact on testing efficacy, accuracy, and interpretation are all prominent topics. Multiple assays using various methodologies have been developed and implemented in the clinical diagnostic laboratory setting. At UC Davis Health we primarily utilize molecular methods, namely reverse transcriptase (RT) polymerase chain reaction (PCR) assays, for detection of the virus but point of care testing as well as serologic testing is available [1]. Serologic assays—particularly quantitative serologic tests—are an increasingly significant topic in the assessment of vaccination response and risk of breakthrough infections. Until recently, the serologic tests available at our institution were qualitative assays [1]. The goal of this blog article is to discuss a semi-quantitative serology test recently implemented at UC Davis Health. Information presented below is current at the time of posting this article.
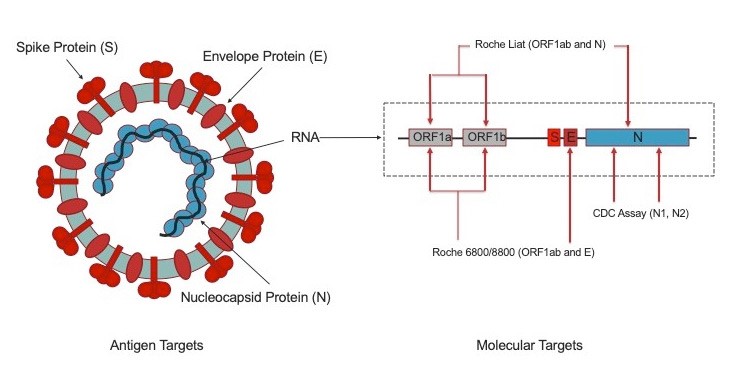
Viral Structural Proteins
SARS-CoV-2 has four structural proteins, nucleocapsid (N), spike (S), membrane (M) and envelope (E) (figure 1) [1,2,3,4]. The S protein consists of two subunits, S1 which contains the Receptor Binding Domain (RBD, the target of neutralizing antibodies) and S2. The nucleocapsid and spike proteins are considered the main immunogens and are widely used in the development of serologic tests or immunoassays [2,3,4].
Serologic Testing
Serology testing measures the host antibody response in the form of immunoglobulins (Ig) such as IgM, IgA, or IgG following infection and/or vaccination. IgM production begins as early as three days post-infection with IgG appearing as early as seven days post-infection. By day 14, the majority of individuals should have a detectable antibody response. False negative serology results may occur if testing is performed too soon after exposure where antibody production is non-detectable (“window period”) [1].
Anti-nucleocapsid serology tests can be used in settings where prior infection with SARS-CoV-2 is suspected [2,3,4]. The nucleocapsid protein is relatively conserved across the various coronaviruses (including non-SARS-CoV-2), making the anti-nucleocapsid serology assays less specific (and prone to false positive results) than their anti-spike assay counterparts. The anti-spike serology assays detect antibodies developed after prior infection but can also detect antibodies developed after vaccination [2,3,4]. However, with the emergence of genetic variants of SARS-CoV-2, concern has arisen over the ability of these assays to detect antibodies to viral variants.
The previous UC Davis serology assay was the LIAISON® SARS-CoV-2 S1/S2 IgG assay, which detected IgG antibodies using a recombinant-expressed spike protein that encompassed both the S1 and S2 domains of the spike protein [1,5]. The S1 subunit is unique in that it also contains the RBD, therefore antibodies against this region are most likely to serve as effective neutralizing antibodies. Studies suggest the LIAISON® SARS-CoV-2 S1/S2 IgG assay correlated well with neutralizing antibody testing [6]. This was a qualitative assay developed for detection of antibodies to SARS-CoV-2 in human serum and plasma. It was not developed with the intention of quantifying the antibodies present [5].
The LIAISON® SARS-CoV-2 TrimericS IgG assay is the 2nd generation of Diasorin’s immunoassay, developed for the semi-quantitative determination of IgG antibodies to the SARS-CoV-2 spike protein [7,8]. The assay uses the complete recombinant-expressed trimeric spike protein. The use of these larger antigen, as opposed to shorter fragments such as those in the previous assay or the commonly used RBD region, provides a larger number of epitopes to capture antibodies, making the assay more sensitive [7,8]. This is useful in the setting of spread of new viral variants, where there was concern for markedly reduced antibody detection in the setting of the variant’s mutations. The assay exhibits a sensitivity of 99.7% and specificity of 99% [8].
The assay is intended as an aid in the diagnosis of COVID-19, but because of its semi-quantitative nature it also has theoretical utility in the study of the immune status of infected and/or vaccinated patients by providing a quantification of IgG antibodies against SARS-CoV-2. In the setting of vaccinated individuals, quantitative serologic testing could offer insight as to who is more susceptible to breakthrough infection after vaccination—an uncommon but important occurrence [9]. These benefits are theoretical however, and the clinical applicability of a semiquantitative result is currently unknown [7].
Results of the LIAISON SARS-CoV-2 TrimericS IgG assay will be reported as a quantitative result in arbitrary units per milliliter (AU/mL) along with a Positive or Negative result. The cutoff for a positive result in our lab is 13 AU/mL, as compared to the cutoff of 15 AU/mL on the S1/S2 IgG assay. The limitations of this assay are similar to those of other immunoassays. Positive SARS-CoV-2 serology results alone are not diagnostic, and as alluded to previously they do not definitively indicate that a patient has had prior SARS-CoV-2 infection [1]. Additionally, not all antibodies produced against a virus, such as SARS-CoV-2, confer immunity [1]. Because semiquantitative SARS-CoV-2 antibody assays are not standardized, and the performance characteristics of each semiquantitative SARS-CoV-2 antibody test is uniquely established, results from different semiquantitative SARS-COV-2 antibody assays are not comparable [1,5,6]. As stated in previous best practice blogs articles, SARS-CoV-2 serology SHOULD NOT be used alone as a COVID-19 diagnostic test without paired molecular diagnostics, to determine immune status post-COVID-19, to guide personal protective equipment use, or for return-to-workplace decisions [1].
References
- Tran, N., Cohen, S., Waldman, S., & May, L. (2021). Review of COVID-19 Testing Methods. Retrieved 22 August 2021.
- Brochot, E., Demey, B., Touzé, A., Belouzard, S., Dubuisson, J., & Schmit, J. et al. (2020). Anti-spike, Anti-nucleocapsid and Neutralizing Antibodies in SARS-CoV-2 Inpatients and Asymptomatic Individuals. Frontiers In Microbiology, 11. doi: 10.3389/fmicb.2020.584251
- MacDonald, A. (2021). COVID-19 Antibody Testing: S vs. N Protein. Retrieved 22 August 2021.
- Rikhtegaran Tehrani, Z., Saadat, S., Saleh, E., Ouyang, X., Constantine, N., & DeVico, A. et al. (2020). Performance of nucleocapsid and spike-based SARS-CoV-2 serologic assays. PLOS ONE, 15(11), e0237828. doi: 10.1371/journal.pone.0237828
- FACT SHEET FOR HEALTHCARE PROVIDERS: DiaSorin Inc. LIAISON® SARS CoV-2 S1/S2 IgG assay. (2021). Retrieved 23 August 2021.
- Muecksch F, Wise H, Batchelor B, et al. Longitudinal serological analysis and neutralizing antibody levels in coronavirus disease 2019 convalescent patients. J Infect Dis 2021;233:389-398
- FACT SHEET FOR HEALTHCARE PROVIDERS: DiaSorin Inc. LIAISON SARS-CoV-2 TrimericS IgG. (2021). Retrieved 23 August 2021.
- Bonelli, F., Blocki, F., Bunnell, T., Chu, E., De La O, A., & Grenache, D. et al. (2021). Evaluation of the automated LIAISON® SARS-CoV-2 TrimericS IgG assay for the detection of circulating antibodies. Clinical Chemistry And Laboratory Medicine (CCLM), 59(8), 1463-1467. doi: 10.1515/cclm-2021-0023.
- Bergwerk, M., Gonen, T., Lustig, Y., Amit, S., Lipsitch, M., & Cohen, C. et al. (2021). Covid-19 Breakthrough Infections in Vaccinated Health Care Workers. New England Journal Of Medicine. doi: 10.1056/nejmoa2109072



