Resident Program - Case of the Month
February 2022 – Presented by Dr. Jasper X. Zheng (Mentored by Dr. Elham Kamangar)
Discussion
Introduction:
Anastomosing hemangioma (AH) is a rare benign vascular entity that occur predominantly in the genitourinary tract and deep soft tissues of older adults. The name is derived from the characteristic anastomosing sinusoidal capillary-sized vessels that constitute this lesion [1,2].
Clinical Presentation:
Initially it was thought to be limited to the genitourinary system. Since its initial documentation by Montgomery and Epstein in 2009, approximately 120 cases have been reported in literature with the most common site of occurrence located in the kidneys and retroperitoneal adipose tissue [2-6]. It can also be found in testis, spermatic cord, ovaries, adrenals, liver, small bowel, colon, mesentery, bladder, soft tissues, bones, and skin [2-6]. The usual age of presentation is around mid to late forties for renal anastomosing hemangiomas and mid-sixties for nonrenal anastomosing hemangiomas [2-5]. The most common clinical presentation of anastomosing hemangiomas, whether it is of renal or nonrenal origin, is an asymptomatic incidentally discovered mass on imaging [2]. At a certain size, anastomosing hemangiomas are capable of causing mass effect, localized pain, and organ dysfunction [2, 6]. In terms of prognosis, anastomosing hemangiomas, even with multicentric or infiltrative pattern, are still benign [2, 6].
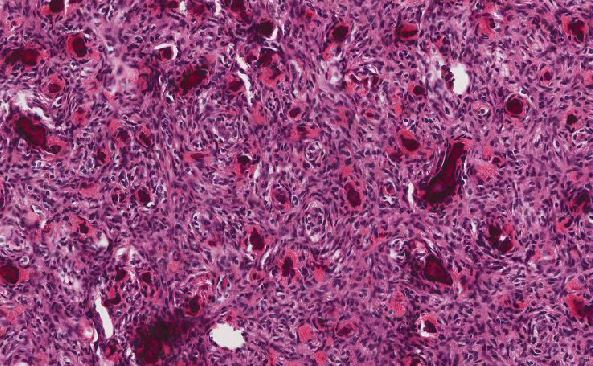
Macro and Microscopic Features:
Grossly, anastomosing hemangiomas is typically a well-circumscribed, frequently unencapsulated tumor with hemorrhagic mahogany colored cut surface and spongy consistency [2,6].
Histologically, anastomosing hemangiomas is composed of loosely lobulated neoplasm of anastomosing sinusoidal capillary sized vessels with scattered hobnail endothelial cells [1-6]. Mitosis is rare or absent. Mild focal cytological atypia is a common finding; however, multilayering of endothelial cells are always absent [2, 6]. Focal areas of central sclerosis, necrosis, and vascular thrombi are usual histological findings in anastomosing hemangiomas [2,6]. The desirable features for diagnosis of this entity are extramedullary hematopoiesis and some hyaline globules [2,6].
Genetic Profile:
The notable genetic feature of anastomosing hemangiomas is the presence of recurrent GNAQ and GNA14 mutations [4, 5]. The alterations of Gaq family serves as a common pathway for the pathogenesis of benign small vessel lesions and more importantly, it is absent in angiosarcoma [4,5].
Differential Diagnosis:
Anastomosing hemangioma is most easily confused with well differentiated angiosarcoma. To distinguish the two, angiosarcomas have diffusely infiltrative growth pattern with irregular and poorly formed vascular channels that is lined by hyperchromatic, mitotically active, and multilayered endothelial cells [2,6]. Anastomosing hemangioma may grow irregularly into surrounding soft tissue or viscera, but it lacks the diffusely infiltrative growth pattern. Anastomosing hemangioma may exhibit mild nuclear atypia, but it lacks the malignant features such as hyperchromasia, increased mitotic activity, and endothelial multilayering. Distinguishing these two enties is imperative to prevent unnecessary damaging over treatment.
Other possible differential diagnosis presented here in table format:
Distinguishing Histological Features |
Distinguishing IHC Staining |
Genetic Profile |
|
Anastomosing hemangioma |
scattered hobnail endothelial cells, mild atypia, extramedullary hematopoiesis, hyaline globules | Positive for CD31, CD34, FLI1, FVIII, ERG Negative for CD8, D2-40, HHV8 |
GNAQ and GNA14 mutations |
Angiosarcoma, well differentiated |
hyperchromatic cells, endothelial cell multilayering, significant mitotic activity, diffusely infiltrative growth pattern | Positive for CD31, CD34, FLI1, ERG | Negative for GNAQ mutation CIC Gene Abnormalities |
Retiform hemangioendothelioma |
long, branching vascular channels (rete testis like), lined by hyperchromatic hobnailed endothelial cells | Positive for CD31, CD34, ERG | YAP1 and MAML2 gene abnormalities |
Hobnail hemangiomas |
grow in a biphasic pattern, with dilated, superficial vessels lined by slightly prominent “hobnail” endothelial cells and a deeper dermal proliferation of capillaries forming slit-like spaces | Positive for CD31, CD34, ERG, D2-40 | NA |
Kaposi sarcoma |
Slitlike vascular spaces, infiltrative pattern, hyalin globules, plasma cells | Positive for HHV8 and D2-40 | NA |
Splenosis |
Occur secondary to previous splenic trauma. Normal capsule, red and white pulp, and pencillar arterioles | Positive for CD8 (Red pulp) | NA |
References
- Montgomery E, Epstein JI. Anastomosing hemangioma of the genitourinary tract: a lesion mimicking angiosarcoma. Am J Surg Pathol. 2009 Sep;33(9):1364-9. doi: 10.1097/PAS.0b013e3181ad30a7. PMID: 19606014.
- Lappa E, Drakos E. Anastomosing Hemangioma: Short Review of a Benign Mimicker of Angiosarcoma. Arch Pathol Lab Med. 2020 Feb;144(2):240-244. doi: 10.5858/arpa.2018-0264-RS. Epub 2019 Apr 8. PMID: 30958692.
- John I, Folpe AL. Anastomosing Hemangiomas Arising in Unusual Locations: A Clinicopathologic Study of 17 Soft Tissue Cases Showing a Predilection for the Paraspinal Region. Am J Surg Pathol. 2016 Aug;40(8):1084-9. doi: 10.1097/PAS.0000000000000627. PMID: 26945338.
- Bean GR, Joseph NM, Gill RM, Folpe AL, Horvai AE, Umetsu SE. Recurrent GNAQ mutations in anastomosing hemangiomas. Mod Pathol. 2017 May;30(5):722-727. doi: 10.1038/modpathol.2016.234. Epub 2017 Jan 13. PMID: 28084343.
- Bean GR, Joseph NM, Folpe AL, Horvai AE, Umetsu SE. Recurrent GNA14 mutations in anastomosing haemangiomas. Histopathology. 2018 Aug;73(2):354-357. doi: 10.1111/his.13519. Epub 2018 May 21. PMID: 29574926.
- WHO Classification of Tumours Editorial Board, Soft Tissue and Bone tumours, Lyon (France): International Agency for Research on Cancer; 5th Edition, Volume 3, 2020.
- Huang SC, Zhang L, Sung YS, et al. Recurrent CIC Gene Abnormalities in Angiosarcomas: A Molecular Study of 120 Cases With Concurrent Investigation of PLCG1, KDR, MYC, and FLT4 Gene Alterations. Am J Surg Pathol. 2016;40(5):645-655. doi:10.1097/PAS.0000000000000582
- Antonescu, Cristina R. MD*; Dickson, Brendan C. MD†; Sung, Yun-Shao MSc*; Zhang, Lei MD*; Suurmeijer, Albert J.H. MD‡; Stenzinger, Albrecht MD§; Mechtersheimer, Gunhild MD§; Fletcher, Christopher D.M. MD∥ Recurrent YAP1 and MAML2 Gene Rearrangements in Retiform and Composite Hemangioendothelioma, The American Journal of Surgical Pathology: December 2020 - Volume 44 - Issue 12 - p 1677-1684 doi: 10.1097/PAS.0000000000001575

 Meet our Residency Program Director
Meet our Residency Program Director