Learn how the Department of Pathology and Laboratory Medicine has worked to address the COVID-19 pandemic
Laboratory testing conducted in our department is key to identifying COVID-19 infection, monitoring disease and response to treatment, and ensuring health. Our research discovers new knowledge that is critical to improving understanding of this novel virus and helps us create new and better approaches and tools, such as more accurate tests and effective therapies and vaccines.
Here are some of our many contributions that are helping the world learn how to address this new disease.
 Established our own COVID-19 testing capabilities
Established our own COVID-19 testing capabilities
We set up and validated our own COVID-19 testing in less than three weeks and used it to help others in the region.
Organizations interested in arranging COVID-19 testing from our laboratory can submit a formal request at this link: https://ucdavis.health/COVIDLab
Our laboratory was also one of the first in the nation to introduce a rapid combined COVID-19 and flue test.
 Expanding testing across California
Expanding testing across California
UC Davis and UC San Diego have partnered to create COVID-19 testing hubs throughout the state of California. Department faculty have helped transform a research lab into a pop-up clinical laboratory.
 Testing and validating novel vaccines
Testing and validating novel vaccines
Pre-clinical testing of immune response from VaxiPatch, a single-dose dermal patch with a metal microneedle array to deliver vaccines against SARS-CoV-2, is taking place at the Mouse Biology Program under the guidance of the lead veterinarian and scientist who is a member of our department.
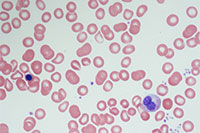 Sharing unique clinical findings
Sharing unique clinical findings
Our hematopathology team describe unusual hematologic findings in the first community-acquired COVID-19 infection in a report published in the American Journal of Hematology.
 Using Our Research Expertise to Create Better Tests
Using Our Research Expertise to Create Better Tests
Through our Center for Diagnostic Innovation, we are working with industry partners to create a new non-PCR test for COVID-19 that uses mass spectroscopy and artificial intelligence. This new test is faster and less expensive than existing tests. Multi-site clinical trials for FDA approvals are in progress.



