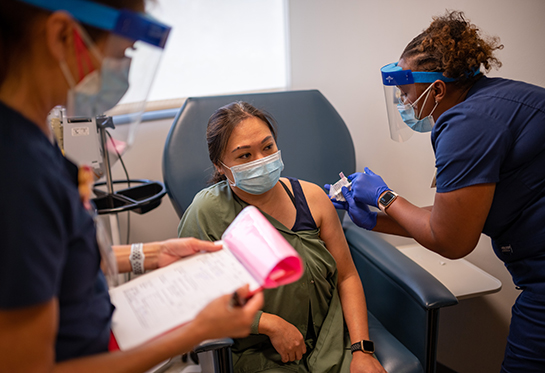
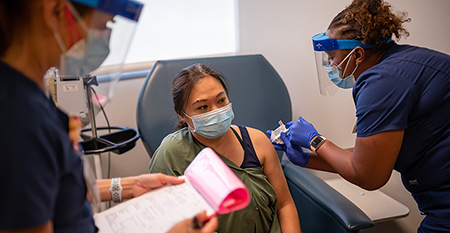
UC Davis School of Medicine tests potential COVID-19 treatments and vaccines
19 months and nearly 43 million cases after the first confirmed COVID-19 infection in the U.S., the search for safe and effective treatments continues at the UC Davis School of Medicine.
As the pandemic spread around the globe, researchers at the School of Medicine partnered with drug developers and funding agencies to find and test potential therapies and vaccines to SARS-CoV-2, the virus that causes COVID-19.
"UC Davis School of Medicine, renowned for its research expertise, is a trusted partner on many COVID-19 clinical trials and research studies. We are immensely grateful to all our researchers who continue to be at the forefront of efforts to cure this terrible disease," said Allison Brashear, the dean of the UC Davis School of Medicine.
UC Davis received about $42 million in COVID-19 research funds in 2020-21. The School of Medicine’s share was more than two-thirds of that, nearly $28.5 million.
With more than 75 COVID-19 research grants, the School Medicine conducted at least 28 studies to find potent treatments and vaccines.
Ongoing clinical trials for potential COVID-19 treatments
UC Davis School of Medicine researchers are currently recruiting for multiple clinical trials, including those using stem-cell-based therapies, monoclonal antibodies, and other experimental drugs. These include:
- Adjustment on Immune Response to COVID-19 Vaccination in Kidney Transplant Recipients (ADIVKT): This study looks at the effect of immunosuppression reduction on antibody response to a third dose of mRNA COVID vaccine in kidney transplant patients.
- Capricor's IV drug: This study tests how well intravenous infusions of CAP-1002 work in hospitalized patients with coronavirus. CAP-1002, based on cardiac stem cell tissues, could decrease inflammation, block or prevent tissue scarring, and stimulate new tissue growth.
- Remdesivir and other medications (I-SPY COVID): This study seeks to determine if the antiviral drug remdesivir used alone or in combination with other drugs can treat COVID-19 and reduce the recovery time for those with severe symptoms.
- ACTIV-3: This publicly funded study tests several agents against COVID, among them, AstraZeneca’s experimental antibody (AZD7442) and Pfizer’s antiviral drug (PF-07304814) to see if these can reduce the severity of the disease and shorten hospital stays for people with COVID-19.
- Selinexor (KPT-330): This study is investigating whether Selinexor, an FDA-approved drug for treating multiple myeloma, speeds up the recovery of COVID-19 patients and reduces their viral load, hospital stay, and complications or chance of dying, compared to standard of care treatment.
- Regeneron's monoclonal antibody cocktail: This study is looking at Regeneron's combined monoclonal antibodies (REGN10933 + REGN10987) as a treatment for non-hospitalized patients with COVID-19. Monoclonal antibodies are a special kind of manufactured antibodies that restore, enhance, or mimic the natural immune system. These antibodies are also being tested for household members of patients with COVID-19.
- Isavuconazole to prevent COVID-19-associated pulmonary aspergillosis: This study is to find out more about the prevention of fungal infections, specifically aspergillosis, in people with COVID-19 infection. Researchers are testing whether the drug isavuconazole can decrease the time COVID patients stay in the ICU and the hospital.
- Experimental stem cells for the treatment of acute respiratory distress syndrome (ARDS): This publicly-funded trial aims to see how effective mesenchymal stromal cells – a type of stem cells - are in reducing inflammation associated with ARDS and in helping damaged lungs repair themselves. ARDS is one of the severe complications of COVID-19 infection.
The researchers also recruited for multiple clinical trials, including:
- Molnupiravir (MK-4482): The study tests an experimental antiviral drug called molnupiravir (MK-4482) in patients with COVID-19. Molnupiravir, by Ridgeback Biotherapeutics and Merck & Co, is taken orally, making it easier to use and distribute.
- Otilimab in severe COVID-19 related disease (OSCAR): This trial assesses the efficacy and safety of GSK's otilimab drug for the treatment of severe pulmonary COVID-19 related disease.
- Cell therapy to treat patients with COVID-19: This study tests how well a treatment with natural killer cells (CYNK-001) works in people with COVID-19. Natural killer cells from the human placenta are immune cells known to kill some types of cancers without hurting normal healthy tissues.
- Regeneron’s monoclonal antibody cocktail: This study tested Regeneron’s combined monoclonal antibodies as a treatment of hospitalized patients with COVID-19.
- Pfizer IV drug in hospitalized patients: The study tests the safety and efficacy of the PF-07304814 drug in hospitalized patients with mild to moderate COVID-19.
- AT-527: This study tests the efficacy and side effects of AT-527 drug, an oral antiviral experimental drug, in hospitalized people with COVID-19.
- Intramuscular injections of PLX-PAD: The study evaluates injections of stromal cells (called PLX-PAD) in the muscle for the treatment of severe COVID-19 patients. It tests if PLX-PAD can help patients intubated and on ventilators due to COVID-19 recover faster with fewer complications. PLX-PAD cells, extracted from the human placenta, have regenerative potential that might help reduce tissue damage caused by hyperimmune reaction to coronavirus.
COVID-19 vaccine testing
The UC Davis School of Medicine has also taken part in testing potential COVID-19 vaccines, including the now FDA-approved Pfizer vaccine for adults and the under-review Novavax vaccine for adults and children (12-17 years old).
Currently, researchers are testing a booster COVID-19 Pfizer shot to adults. They are also assessing SARS CoV-2 infection, viral shedding and potential transmission in young adults immunized with the Moderna's vaccine.
Developing rapid COVID-19 tests

With the advent of the pandemic, clinical lab scientists at UC Davis Department of Pathology and Laboratory Medicine developed an in-house COVID-19 test. Their remarkable achievement was a testament to a great interdisciplinary teamwork culture spanning across many departments.
One notable collaboration between UC Davis School of Medicine and the industry is the development of a rapid COVID-19 test. The new test uses an analytical instrument known as a mass spectrometer, which is paired with a powerful machine-learning platform to detect SARS-CoV-2 in nasal swabs.
Other COVID-19-related studies at UC Davis School of Medicine
Researchers are also conducting studies to better understand COVID-19 and how it impacts people and their health. Some of these studies include:
- COVID-19 survey for workers: This evaluates the impacts of the COVID-19 pandemic and its related lockdown measures on workers throughout the U.S.
- Total-body parametric 18F-FDG PET of COVID-19: This study measures the differences between the PET/CT scans of COVID-19 patients and healthyl subjects.
- Serologic response of COVID-19 patients: This study seeks to determine how well the antibody blood tests work and how the body's immunity responds to the infection.
- Characterization of multisystem inflammatory syndrome in children (CHARMS) and its relationship to Kawasaki disease: This research is trying to understand how the body responds to inflammation caused by Kawasaki disease and to the coronavirus, including the Multisystem Inflammatory Syndrome in Children (MIS-C).
- Presence of SARS-CoV-2 in infant stool: The purpose of this study is to see how long the coronavirus is found in infant feces after infection.
- Experimental PET/CT Radiotracer after COVID-19 infection: This study tests the ability of a new UC Davis developed drug called "radiotracer" to detect and locate possible lung damage caused by COVID-19.
- Characterizing T cell behavior in patients recovering from COVID 19: This research aims to understand the ways "radiotracer" can help study how individuals recover from COVID-19.
- COVID-19 in cancer patients: This study is to understand how COVID-19 affects the outcomes of patients undergoing cancer treatment and how having cancer affects COVID-19.
- The “BLUE CORAL” study collects health information about COVID-19 from hospitalized patients with coronavirus to better understand how to treat COVID-19.
"We take this opportunity to thank all of the participants who volunteer in our clinical trials," said Timothy Albertson, chair of internal medicine at UC Davis School of Medicine and principal investigator on multiple COVID-19 clinical trials. "Their contribution helps to save lives and push science forward."
Learn more about how to sign up for UC Davis Health clinical trials or studies here.