Study explains why food high in saturated fat may lead to plaque build-up in arteries
Research also points to a drug that could block the negative effects of a high-fat diet
We all know that fatty foods are linked to blocked arteries and a higher risk for heart disease, but why or how the food becomes toxic has remained a mystery, until now. New research involving UC Davis Health may provide an answer.

The study, published Aug. 13 in Science, suggests that consuming food rich in saturated fat and choline - a nutrient found in red meat, eggs and dairy products - increases the number of metabolites that build plaques in the arteries. It also points to a possible drug that can block the effects of the high-fat diet on the gut and the arteries.
“Our study shows how a high-fat diet disrupts the bacterial balance in the gut and leads to the production of harmful substances implicated in cardiovascular diseases,” said Andreas Bäumler, professor of medical microbiology and immunology at UC Davis Health and co-lead author on the study.
The gut-heart connection
A healthy gut has an anaerobic environment suitable for good bacteria. The mitochondria in the cells of the intestinal lining serve as the power engines burning oxygen. Previous studies have shown that a high-fat diet disrupts the work of the mitochondria in the gut. This disruption shuts the engine’s ability to burn oxygen, leading to oxygen leakage into the intestine.
Higher oxygen levels in the gut promote bacterial imbalances. The good bacteria get replaced by harmful microbes, such as E-coli, that are more oxygen tolerant.
“It was known that exposure to a high-fat diet causes dysbiosis — an imbalance in the microbiota favoring harmful microbes, but we didn’t know why or how this was happening,” said Mariana X. Byndloss, co-lead author and assistant professor of Pathology, Microbiology and Immunology at Vanderbilt University Medical Center.
Choline is a common component of a Western-style high-fat diet. When bacteria in the gut break down food containing choline, they produce a metabolite known as trimethylamine (TMA). TMA is made in the gut but oxidized and converted in the liver into TMAO (trimethylamine-N-oxide).
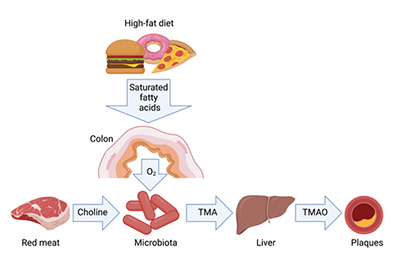
The study showed that the microbiota does not break down choline when it is consumed with a low-fat diet. But saturated fatty acids present in a high-fat diet weaken the intestinal lining’s ability to balance the microbiota and prevent oxygen leakage into the intestine.
“The increased oxygen availability resulting from a high-fat diet escalates microbial metabolization of choline, leading to elevated TMAO levels in the blood,” said Bäumler. “This, in turn, creates more risk for heart disease and plaque deposits in arteries.”
Many studies have linked TMAO to atherosclerosis, a cardiovascular disease in which fatty deposits build up inside the arteries. People with higher levels of TMAO in their blood may have more than twice the risk of serious heart problems, compared with people who have lower levels.
A drug may boost the cell’s power engines and block high-fat diet effect
The study found that 5-aminosalicylic acid (5-ASA), a type of drug that strengthens the ability of the intestinal lining to withhold oxygen, was able to prevent the microbiota from producing harmful metabolites from a Western-style high-fat diet. The 5-ASA activated mitochondrial bioenergetics specifically in the intestinal epithelium.
“This is evidence that it’s possible to prevent the negative outcomes associated with a high-fat diet,” Byndloss said. A drug such as 5-aminosalicylic acid might be used in conjunction with a probiotic to both restore a healthy intestinal environment and boost beneficial microbe levels, she added.
Mesalazine, a 5-ASA drug, could restart the energy factories in the intestinal lining and prevent oxygen and nitrate from residing in the gut. It may neutralize the harmful effects of a high-fat diet and prevents the microbiota from producing toxic levels of TMA metabolites.
Co-authors on this study are Woongjae Yoo, Jacob Zieba, Nora Foegeding, Teresa Torres, Catherine Shelton, Nicolas Shealy, Austin Byndloss, Stephanie Cevallos, Erik Gertz, Connor Tiffany, Julia Thomas, Yael Litvak, Henry Nguyen, Erin Olsan, Brian Bennett, Jeffrey Rathmell and Amy Major.
The research was supported by the V Foundation for Cancer Research, American Cancer Society, United States-Israel Binational Science Foundation, USDA/NIFA, Crohn’s and Colitis Foundation, and the National Institutes of Health (grants DK058404, CA236733, TR002243, AI0441780, AI096528, AI112445, AI112949, AI146432, AI153069).
Yoo et al. (2021). High-fat diet-induced colonocyte dysfunction escalates microbiota-derived trimethylamine-N-oxide, Science, DOI:10.1126/science.aba3683